 
Movie depicting the 4 d z 2 electron density function ( ψ 4 d z 2) 2. In other words, there is a maximum probability of finding an electron in an atomic orbital. All values of electron density are of necessity not negative since the square of any real number cannot be less than zero. An atomic orbital is a three-dimensional description of the location of an electron around the nucleus of an atom. Answer : The answer is 1 radial node The number of radial nodes is related to the principal quantum. By choosing different values of electron density, denoted by the bar moving up and down on the line plot or by the moving plane on the surface plot, then the size of the three-dimensional plot changes. This 4d orbital is one of four four-lobed orbitals (called 4dxy, 4dyz, 4dxz and 4dx2-y2) arranged around the atom. How many radial nodes are present in 4d orbital. 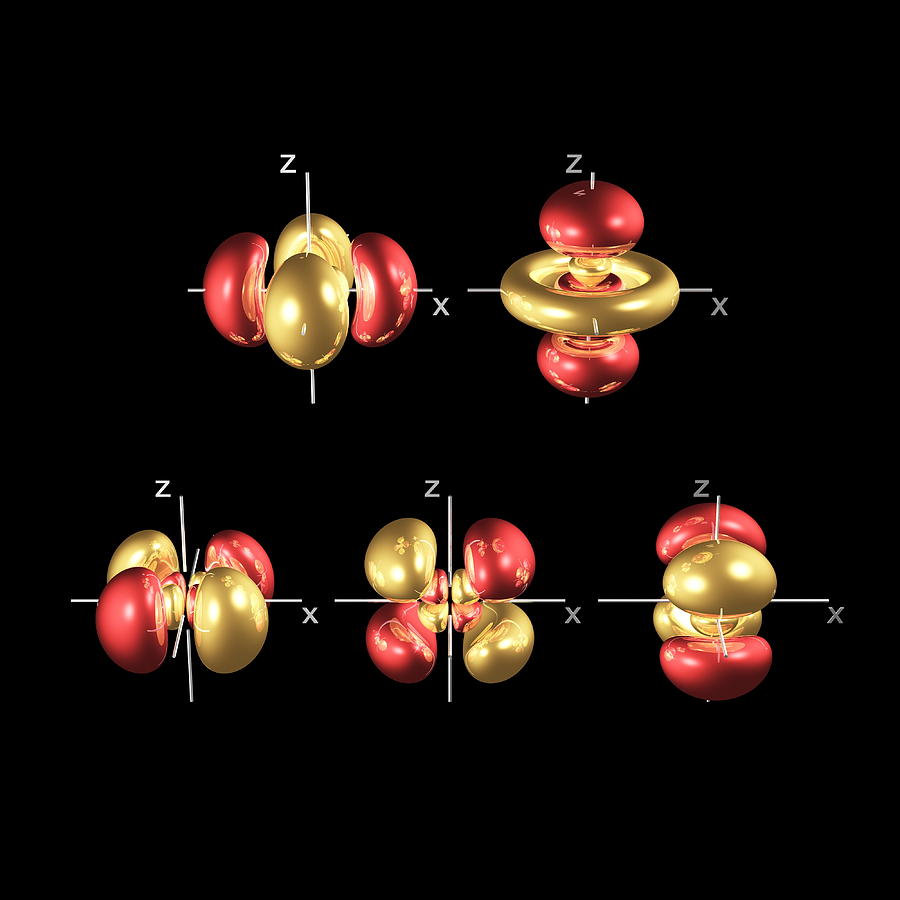
The "surface" of the three-dimensional orbital at the top centre of each movie represents points for which the electron density for that orbital is the same - an isosurface. See the 4 d wave function page for movies depicting the 4 d the wave functions and nodal structures. A wave function for an electron in an atom is called an atomic orbital this atomic. In all cases the red zones are where the 4 d wave function has positive values and the white zones denote negative values. This page contains movies depicting the 4 d xy, 4 d xz, 4 d yz, and 4 d x 2- y 2 wave functions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
